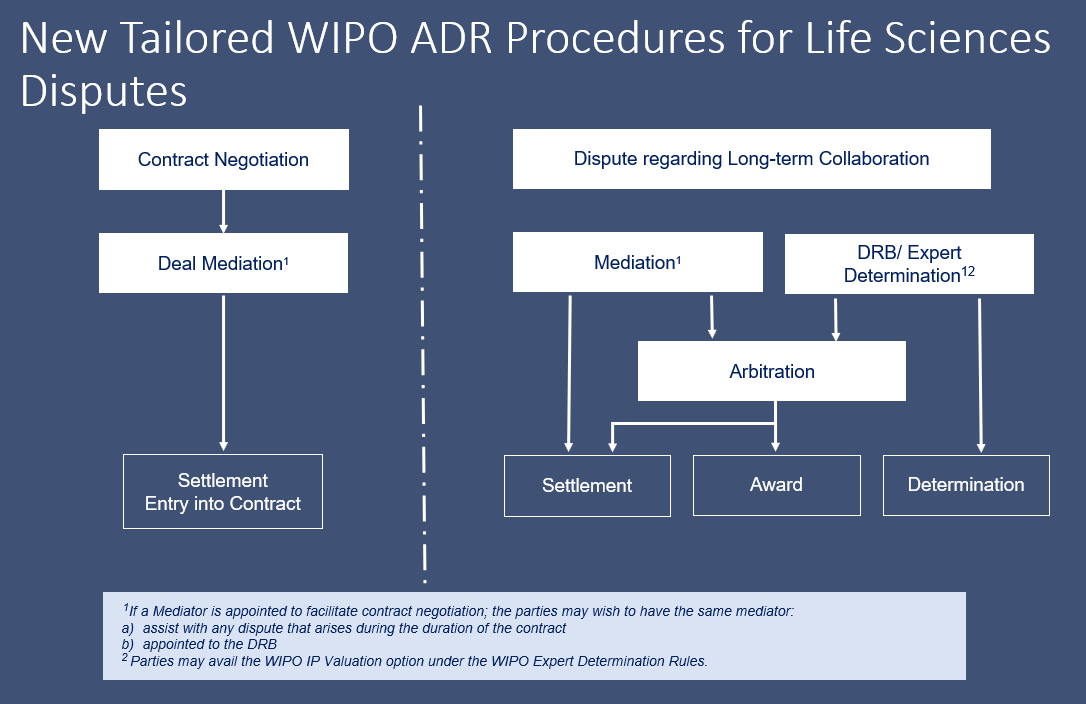
The WIPO Center provides dispute resolution advice and case administration services to facilitate contract negotiations between parties (Deal Mediation) or to help parties resolve disputes in the area of life sciences.
Life Sciences Disputes
- Fifteen percent (15%) of the arbitration and mediation cases filed with the WIPO Center relate to life sciences. The life sciences caseload also includes Deal Mediation, where parties adapt WIPO Mediation to facilitate contract negotiations.
- The subject matter of WIPO life sciences ADR cases includes contractual and non-contractual/infringement disputes. These disputes relate to patents, designs, trademarks, or know-how/data. Contractual disputes arise out of R&D agreements, joint development agreements, options and licensing agreements, manufacturing, marketing and distribution agreements.
- WIPO life sciences cases are normally international, with amounts in dispute between USD 50,000 and up to USD 1 billion.
- Parties involved in these disputes include originator and generic pharmaceutical companies, diagnostics and medical device companies, start-ups, research institutions, and universities, from Asia, Europe and North America.
The WIPO Center makes available examples of WIPO mediations and WIPO arbitrations in life sciences.
As time- and cost-efficient alternatives to court litigation, alternative dispute resolution (ADR) mechanisms such as mediation, arbitration, or Dispute Resolution Boards, offer parties and their lawyers the opportunity to adopt practical and satisfactory solutions. ADR allows parties to choose a mediator, arbitrator or expert knowledgeable in the specific legal field, life science sector, IP and dispute resolution. It provides a neutral forum for resolving disputes through a procedure that can take into account parties’ business, research and other strategic goals.
- biodiversity
- biotechnology
- copyright
- counterfeiting
- design
- financing
- IP infringement
- IP licensing
- IP valuation
- know-how
- marketing authorizations
- material transfer agreements
- mergers & acquisitions
- misleading advertising
- patents packaging
- pharmaceuticals
- product liability
- product piracy
- research and development agreements
- royalties
- settlement agreements
- SPCs (supplementary protection certificates)
- software agreements (e.g. bioinformatics)
- technology transfer
- trademarks
- unfair competition
- associations
- biotech companies
- funding bodies
- generic companies
- government agencies
- insurance companies
- IP asset management agencies
- medical devices and diagnostics companies
- originator companies
- patent marketing agencies
- research institutions
- insurance companies
- start-up companies
- technology transfer offices
- universities
- vehicles designed to acquire, transfer or assign IP rights
WIPO ADR Services for Life Sciences
The WIPO Mediation, (Expedited) Arbitration and Expert Determination Rules are well suited for life sciences disputes. In addition, as part of WIPO’s COVID-19 Related Services and Support, the WIPO Center offers tailored WIPO ADR options to facilitate contract negotiation and dispute management, set out in further detail below: