6.1 Overview of the patent system
6.1.1 Evolution of the patent system
Intellectual property (IP) rights are governed by national law, which for members of the World Trade Organization (WTO), shall be in conformity with the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS Agreement).1 The TRIPS Agreement sets out the objective of IP rights in Article 7:
The protection and enforcement of intellectual property rights should contribute to the promotion of technological innovation and to the transfer and dissemination of technology, to the mutual advantage of producers and users of technological knowledge and in a manner conducive to social and economic welfare, and to a balance of rights and obligations.
As a member-nation of the WTO, India was required to amend or enact laws to conform to the TRIPS Agreement. However, this was a challenge for India. A significant reason was that, unlike many other countries, such as the United States of America (U.S.), where the Constitution recognizes the promotion and progress of science and arts and secures exclusivity granted to authors and inventors, the Constitution of India only encourages Indian citizens to have a scientific temper and prescribes a duty to develop the spirit of inquiry and reform.2 The Constitution of India mandates that no one shall be deprived of “property” except with the authority of law.3 Since patents are “property,” there is a positive constitutional entitlement to the grant and recognition of patents. The non-enforceable – but critical – chapter of the “Directive Principles of State Policy” in the Constitution of India further directs the Government to ensure the promotion of public health,4 the reduction of inequalities5 and the securing of systems that ensure ownership and control of resources for the common good.6 The basis and limitations for IP rights are, therefore, the right to property, the directive principles of state policy and the fundamental duties of citizens, apart from the various laws enacted periodically.
The journey of the Indian patent regime is reflected in three different periods: colonization, post-independence and globalization.7
Colonization. India inherited its patent regime from the British rule. When the British colonization of India ended, the Indian Patents and Designs Act, 1911, was in force and had created a system of patent administration in India under an administrative office – the Controller of Patents and Designs.
Post-independence. India enacted its first independent patent law in 1970. It came in the backdrop of two committees constituted to make recommendations: the Bakshi Tekchand Committee in 1949 and, later, the Justice Rajagopal Ayyangar Committee. Focusing on the special socioeconomic conditions in India, the recommendations of these two committees resulted in far-reaching changes in patent laws. Some of the significant changes introduced were with respect to food and drug patents, compulsory licensing, and connected working requirements. The law enacted in 1970 is credited with the growth of various industries, including the pharmaceutical industry, which, in two decades, gave India the distinction of being called “the pharmacy of the world” as Indian drug companies began exporting reasonably priced medicines to many countries.
Globalization. In 1991, India liberalized its economy and adhered to the General Agreement on Tariffs and Trade (GATT 1947), which was succeeded by the WTO, resulting in amendments being introduced in line with the TRIPS Agreement. These amendments saw India bring about fundamental changes permitting product patents in food, medicines and agrochemicals. The flexibilities in the TRIPS Agreement were used to maintain a balance: ensuring that the amendments would be gradually made systemic rather than forcing the closure of already-functioning industries. Statutory provisions relating to chemical and drug patents, patentability and other aspects of the amendments were tested repeatedly in the courts and were upheld as being within the Constitutional scheme while being fully compliant with the TRIPS Agreement. The judgment of the Supreme Court in Novartis v. Union of India8 recognized the need to curb the “evergreening” of patents while acknowledging the need to grant patent protection to incremental innovations. After Novartis, Indian courts have granted interim injunctions to protect patentees’ rights in pharmaceutical9 and agrochemical inventions.10 The courts have also protected claims to standard-essential patents (SEPs) by granting interim injunctions to secure the patentee’s right to royalties even pending trial.11 Courts have granted permanent injunctions12 and damages (in quite significant amounts)13 in cases of patent infringement and have also denied interim injunctions in appropriate cases.14 Each case has been decided on its own facts on the basis of settled legal principles. A current review of decisions would show no pro- or anti-patentee bias in the adjudication of patent cases.
6.1.2 The Justice N Rajagopala Ayyangar Committee Report
In 1957, the Government of India appointed a committee led by a distinguished retired Justice of the Supreme Court of India, Justice N Rajagopala Ayyangar, to examine the revision of the Patents Act and advise the Government in this respect.
The Justice N Rajagopala Ayyangar Committee report stated, in no uncertain terms, that the patent system was a quid pro quo system: the monopoly that a patentee obtains is only in exchange for the disclosure of the invention to the public, free to be used after the monopoly period is over. The quid pro quo, according to the report, also included the obligation on the part of the patentee to work the invention in India. The report also underscored, rather emphatically, that the patent system had failed in India because it had failed to spark the kind of innovation that it sought to encourage – underdeveloped countries could not yield the same result from the patent system as their more developed counterparts could. The patent system was recommended to be continued only because there was no better alternative to achieve better results – in their form at the time, patents were the lesser evil. The report was unequivocal in its apprehension that foreign patentees could misuse the patent system to capture large markets in India at the cost of domestic innovation while simultaneously not investing in the manufacture of the patented product.
The committee’s recommendations were a catalyst for wide changes in Indian patent law, eventually leading to the Patents Act of 1970, replacing the Indian Patents and Designs Act, 1911. The Patents Bill was introduced in 1965 and amended in 1967. The Patents Act, 1970, and Patents Rules, 1972 came into force on April 20, 1972.
6.1.3 The Patents Act, 1970 (pre–TRIPS Agreement)
The Patents Act, 1970, incorporated major provisions to reduce the social costs of foreign-owned patents. It prohibited patents on products useful as medicines and food, shortened the term of chemical process patents, and significantly expanded the availability of compulsory licensing. This spawned a powerful Indian pharmaceutical generic drugs industry.
In Bishwanath Prasad Radhey Shyam v. HM Industries,15 deciding an appeal in a case for infringement of a patent called “Means for Holding Utensils for Turning Purposes,” the Supreme Court said:
The object of the patent law is to encourage scientific research, new technology and industrial progress. Grant of exclusive privilege to own, use or sell the method or the product patented for the limited period, stimulates new inventions of commercial utility. The price of the grant of the monopoly is the disclosure of the invention at the Patent Office, which after the expiry of the fixed period of the monopoly passes into public domain.
The salient features of the Act (as enacted) were:
-
the reduction of the term of patent from 16 to 14 years;
-
a maximum of seven years for the term of a patent for the processes for drugs and foods;
-
no product patents available for food, drugs and medicines, including the products produced or obtained by chemical processes;
-
provisions prescribing nonworking as a ground for the grant of compulsory licenses, licenses of right and the revocation of patents;
-
the empowerment of government to use inventions for its own use;
-
provisions for the use of inventions for government purposes, research or instruction to pupils;
-
the endorsement of a “license of right” to patents related to drugs, foods and products of chemical reactions;
-
the codification of certain inventions as non-patentable;
-
the expansion of the grounds for opposition to the grant of a patent;
-
exemption from anticipation in respect of certain categories of prior publication, prior communication and prior use;
-
provisions for the secrecy of inventions relevant for defense purposes;
-
the mandatory furnishing of information regarding foreign applications;
-
the prevention of abuse of patent rights by voiding restrictive conditions in license agreements and contracts;
-
a provision for appeal to the High Court from decisions of the Controller General of Patents, Designs and Trade Marks (“the Controller”); and
-
the separation of industrial designs from the law of patents.
However, many provisions changed after the TRIPS Agreement, as discussed in Sections 6.1.4.4.3 to 6.1.4.4.5.
6.1.4 International obligations and commitments
India is a member of the WTO, which came into being on January 1, 1995. The WTO administers the General Agreement on Tariffs and Trade (GATT),16 which is an international agreement among countries to promote free international trade in goods. The WTO is a package deal in that its members must abide by the GATT agreement and a series of other international agreements. One such agreement is the TRIPS Agreement. India is also a member of the Paris Convention for the Protection of Industrial Property,17 the Patent Cooperation Treaty (PCT),18 as well as the Budapest Treaty on the International Recognition of the Deposit of Microorganisms for the Purposes of Patent Procedure.
6.1.4.1 The TRIPS Agreement
TRIPS is one of the most comprehensive multilateral agreements on intellectual property rights.
Section 5 of TRIPS deals with patents. Article 27(1) of TRIPS provides that patents will be available for products or processes of inventions in all fields of technology, provided they are new, involve an inventive step and are capable of industrial application. This was a departure from what the Patents Act, 1970 allowed at the time since no patents were allowed for “substances intended for use, or capable of being used, as food or as medicine or as drug.”19 In such cases, only method or process patents were allowed for such substances.
Article 70(8)–(9) of the TRIPS Agreement stipulates that, during the transition period, a country should provide a mechanism for patent protection for pharmaceutical and agricultural chemical products, including the grant of exclusive marketing rights (EMRs). On July 2, 1995, the U.S. alleged that India had not complied with these provisions. It requested the WTO for dispute consultations. A panel to hear the dispute issued a report on September 5, 1997, finding that India was indeed in violation of these TRIPS Agreement provisions.20 India’s appeal also failed. The appellate body found that, as on January 1, 1995, India was required to have a legal mechanism for patent protection as provided under Article 70(8)–(9) of the TRIPS Agreement.21
In 1997, the European Community requested another dispute consultation on similar grounds. The panel set up in this regard also ruled against India.22 Accordingly, in 1999, India introduced an amendment to comply with these requirements. These, and other amendments of 2002 and 2005, are discussed in Sections 6.1.4.4.3 to 6.1.4.4.5.
6.1.4.2 The Doha Declaration
Prior to the adoption of the TRIPS Agreement, most countries did not grant patents for medicines. This helped keep costs affordable and ensured access to medicines. The introduction of product patents for medicines under the TRIPS agreement was a matter of concern for developing countries and least-developed countries. Increasing the number of product patents for medicines implied that the cost of medications would increase and thwart access to medication.
The TRIPS Agreement required, among other things, that all WTO members introduce product and process patents in all fields of technology. Exceptions in fields related to the fulfillment of basic needs, such as in health, were not recognized or permitted.
In 2001, WTO members adopted a declaration at the WTO Ministerial Conference in Doha, Qatar, stating that it was important to interpret the TRIPS Agreement to support public health by promoting access to medicine and the creation of medicines.23 This was important for developing economies, including India, which had stressed the need to expand public health coverage at low and affordable costs. The Doha Declaration agreed that the TRIPS Agreement did not and should not prevent WTO members from taking measures to protect public health.24
The Doha Declaration recognized that the TRIPS Agreement should be interpreted and implemented in a manner conducive to its members, deploying the flexibilities built into the TRIPS Agreement.25 Consequently, each WTO member was free to determine the grounds on which compulsory licenses were to be granted and what constituted a national emergency or other circumstances of extreme urgency for invoking compulsory licensing provisions.26 The Doha Declaration also recognized that many countries had little or no manufacturing capacity in the pharmaceutical sector and might face difficulties in the effective use of the TRIPS Agreement’s compulsory licensing provisions.27 Pursuant to this, an amendment was accepted in Article 31bis of the TRIPS Agreement, permitting countries to grant compulsory licenses even for export to other countries with insufficient or no manufacturing capacity.
The Doha Declaration also clarified flexibilities for members to adopt an international principle of exhaustion of rights28 in accordance with Article 6 of the TRIPS Agreement.29 The principle of exhaustion means that, once patent holders sell a patented product, they cannot prohibit the subsequent resale of that product, since their rights in respect of that product have been “exhausted” by the act of selling the product. The Doha Declaration reaffirmed that members were free to establish their own regime for such exhaustion to ensure that patent rights did not impede legitimate products entering global supply chains.
6.1.4.3 The Patent Cooperation Treaty
The PCT provides a platform to facilitate the filing of a single international patent application to seek protection across PCT contracting states. This is beneficial for an applicant because, in the traditional system, separate applications for patents had to be made in each jurisdiction across the world. The international search reports and written reports generated by the International Searching Authorities as well as International Preliminary Reports on Patentability (Chapter II) drawn by the International Preliminary Examining Authorities assist the applicant in deciding whether to proceed with the national phase and, if so, in which countries, based on the likelihood of success as per the search report. The PCT system has also resulted in a considerable reduction in costs for applicants.
6.1.4.4 Amendments and implementation in India
6.1.4.4.1 Patent Cooperation Treaty implementation in India
India signed and acceded to the PCT in September 1998, which entered into force in India in December 1998. The provisions relating to applications under the PCT were incorporated under the Patents (Amendment) Act, 2002. Under the Patents Act, 1970, an “international application” was defined as an application made as per the provisions of the PCT.30 Four offices in India (New Delhi, Kolkata, Chennai and Mumbai) and the International Bureau in Geneva, Switzerland were designated as receiving offices for international applications. Section 7 of Act prescribes the form in which an applicant makes an application for its invention and also provides for making a simultaneous application under both the PCT and the Act if a corresponding application has been filed before the Controller in India.31
Chapter III of the Patents Rules, 2003, contains the provisions for filing an international application, the form in which an application is to be made, fees payable to the examining authority, time limits for establishing an international search report and other related rules for applications under the PCT. The term of a patent granted in India for a PCT international application is 20 years from the date of its filing under the PCT.32
6.1.4.4.2 Patent prosecution highway
Apart from the PCT system, several countries and regions have recently created “patent prosecution highways” which provide for accelerated examination, the sharing of search reports and so on, which result in the speedier examination and grant of patents. Such prosecution highways can either be bilateral or multilateral. In India, the first patent prosecution highway was initiated in 2019 by the Indian Patent Office as a bilateral pilot patent prosecution highway program with the Japan Patent Office. Guidelines for the same were published, though the pilot is limited to 100 cases per year, on a first-come-first-served basis. Depending on the evaluation of this pilot highway, long-term patent prosecution highways with one or more patent offices across the country may be a reality.
6.1.4.4.3 The 1999 amendment, post–TRIPS Agreement
Upon coming into effect on January 1, 1995, the TRIPS Agreement provided for transitional periods for WTO members to introduce legislation complying with the obligations under the agreement. India has been a WTO member since January 1995.
For developing countries like India, the deadline for complying with the TRIPS Agreement was the year 2000. Article 65(4) of the TRIPS Agreement provided a special transitional provision for those countries that did not grant product patents. As per this provision, an additional period of five years (i.e., until 2005) on the initial TRIPS Agreement transitional period was permitted for introducing product patent protection.
India needed to provide a means for filing patent applications during the transitional period. The “mailbox provision” allowed applicants to file for patents, thereby establishing filing dates, while at the same time permitting members to defer the granting of product patents. In addition, India also needed to provide EMRs in exchange for permission to delay the grant of product patents until January 1, 2005.
Transitional arrangements were introduced through Section 2 of the Patents (Amendment) Act, 1999, through the insertion of Section 5(2) of the Patents Act, 1970, allowing product patent applications to be filed through a “mailbox,” while Chapter IVA provided for the grant of EMRs if certain conditions were fulfilled
EMRs were introduced as a transitory provision to help developing countries that followed a process patent regime to slowly phase into a product patent regime. In order to bring in transitional measures for the recognition of the TRIPS Agreement obligations, the Patents (Amendment) Act, 1999, introduced a system for the grant of EMRs. This allowed inventors to file early applications for the grant of patents and to establish filing dates so that, when patent protection was ultimately granted, these applications would be considered on the basis of the date of filing or priority dates. These provisions were considered necessary under the TRIPS Agreement,33 pending the initiation of a streamlined process in India for granting product patents relating to drugs, pharmaceutical and agricultural chemical products.
EMRs are applicable where a patent is granted for the same product in another WTO member after 1995 (the date of entry into force of the TRIPS Agreement), provided marketing approval for the product was obtained in such other WTO member. However, EMRs are limited only to pharmaceutical and agricultural chemical products. From a simple dictionary definition of the term, the meaning of “exclusive marketing rights” appears to be very similar to that of patent rights; however, in theory, EMRs prevent others from making or using patented products. The rights holder can indirectly prevent others from marketing products based on such use since they would lack the authorization to do so. Patent protection and EMRs are alternatives to each other and are not used concurrently.
EMRs under the 1999 amendment could only be granted for products intended for or capable of being used as a medicine or drug. For an applicant to have the exclusive right to sell or distribute the product in India, pending the grant or rejection of the application for the product patent, the following conditions needed to be fulfilled:
-
a patent and approval to sell the same invention applied for (on or after January 1, 1995) in another WTO member had been granted after the date of making an application for the product patent;34 or
-
a patent for the method, process or manufacture of the invention applied for (on or after January 1, 1995) and relating to the same product had been granted in India on or after the date of making an application for the product patent;35 and
-
approval to sell or distribute the product had been received from the Central Government.36
EMRs were granted for a period of five years from the date of such approval or until the grant or rejection of the application for the product patent, whichever was earlier.
As per the 1999 amendment, no application for the grant of a product patent could be referred by the Controller to an examiner for making a report until December 31, 2004.37 For the said 10-year period, the applications were kept in a “black box,” a figurative expression for applications pending examination. After this date, the application would be referred to an examiner for a report on whether the claimed invention was within the meaning under Section 3 of the Patents Act, 1970, or whether the invention was such for which no patent could be granted under Section 4 of the Act. If the necessary preconditions were not met, the application would be rejected.38 If the preconditions were fulfilled, the Controller could proceed to consider the application for the grant of a patent.39
The 1999 amendment also included provisions authorizing the Central Government – under expedient circumstances and keeping in mind the public interest at large – to sell or distribute the product by itself or through an authority so empowered in writing.40 Moreover, the Central Government also had the power to direct that the product be sold at a price determined by it after specifying its reasons and the public interest involved.
All suits relating to infringement under Section 24B of the Act would be dealt with in the same manner as suits concerning infringement of patents under Chapter XVIII.
In India, some EMRs were granted relating to medicinal products. Suits for infringement restraining violation of EMR rights were also instituted. However, all EMRs came to an end after the full-scale implementation of the amendments with effect from January 1, 2005. With the introduction of the 2005 amendments, all pending applications for the grant of EMRs were automatically considered as applications for product patents and dealt with accordingly.
6.1.4.4.4 The 2002 amendment, post–TRIPS Agreement
This 2002 amendment to the Patents Act, 1970, was introduced to (a) bring the patent regime in India in line with the TRIPS Agreement; (b) bring the law on patents in line with the increasing development of technological capability of India; (c) provide the necessary safeguards for the protection of public interest and national security; (d) harmonize the procedure for the grant of patents in accordance with the international practices; and (e) make the system more user-friendly.
Some of the salient features of the Patents (Amendment) Act, 2002, were as follows:
-
The term of every patent granted after the commencement of the Patents (Amendment) Act, 2002, was increased to 20 years from the date of filing of the application.41
-
The time for restoration of a lapsed patent was increased to 18 months.42
-
A new definition for “invention” was added: a patent could be for a process or product that was new, involved an inventive step or was capable of industrial application.43
-
A new definition for “inventive step” was added.44
-
The negative list of what were not considered inventions (i.e., non-patentable subject matter) was amended and expanded in light of Article 27(2)–(3) of the TRIPS Agreement.45
-
The concept of a request for the publication of a patent application was introduced.46
-
An onus-of-proof provision was introduced, requiring the defendant to prove that its process for obtaining the product in question was different from the patented process in cases where an identical final product was obtained from such a process.47
-
The chapter on compulsory licensing was substituted with provisions and procedures consistent with the TRIPS Agreement,48 and the provisions relating to the license of rights were omitted.49
-
The Bolar exemption was introduced.50
-
The parallel import of patented products was introduced.51
-
All appeals under the Act were redirected from the High Courts to a specialized tribunal (i.e., the Intellectual Property Appellate Board (IPAB)52 since abolished in 2021).53
-
National security provisions were amended.54
6.1.4.4.5 The 2005 amendment, post–TRIPS Agreement
The amendments of 2005 were introduced to bring Indian patent laws into further compliance with the TRIPS Agreement because the transitional period available to India was ending in 2005. Some of the salient features of the Patents (Amendment) Act, 2005, were as follows:
-
The definition of “inventive step” was amended.55
-
The definition of “new invention” was added.56
-
The definition of “patent” was amended.57
-
The negative list of what were not considered inventions (i.e., non-patentable subject matter) was amended.58
-
The provisions that provided that only the process and not the product itself would be patented in cases of inventions relating to food, drugs and medicines were deleted.59 This ensured that product patent protection was available for all fields.
-
The chapter relating to EMRs was omitted,60 and the provisions relating to it were modified.
-
Two levels of opposition were introduced – pre-grant and post-grant. All grounds available for pre-grant opposition were also made available to interested parties for challenging a patent in post-grant opposition within one year from the date of publication of the grant of patent.61
-
Pursuant to the Doha Declaration, the grounds for seeking compulsory licensing were expanded by adding a provision for the issuance of compulsory licenses for the manufacture and export of patented pharmaceutical products to countries that had insufficient manufacturing capacity in the pharmaceutical sector if that country had allowed such importation by notification.62
-
Jurisdiction for trying revocation petitions to revoke granted patents was shifted from the High Courts to the IPAB with a view to extending its jurisdiction to the revocation of patents.63 This now stands reversed in 2021.64
-
Certain provisions were amended to bring the patent regime in India in line with the PCT, to which India is a signatory.65
6.1.5 Patent application trends
Figure 6.1 shows the total number of patent applications (direct and Patent Cooperation Treaty (PCT) national phase entry) filed in India from 2000 to 2021.
Figure 6.1 Patent applications filed in India, 2000–2021
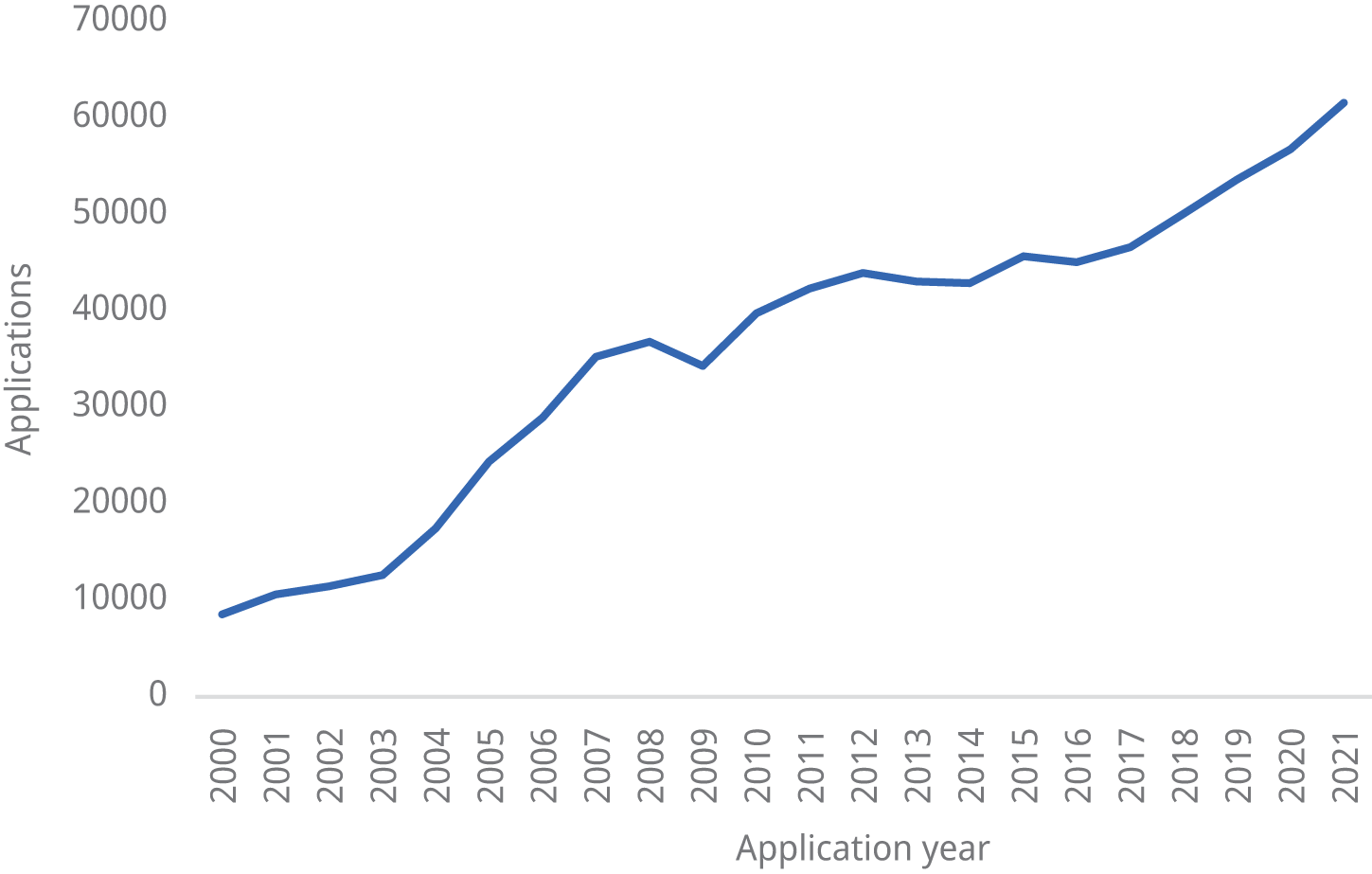
Source: WIPO IP Statistics Data Center, available at www3.wipo.int/ipstats/index.htm?tab=patent
AIR 2013 SC 1311.
E.g., Merck Sharp and Dohme Corp. v. Glenmark Pharmaceuticals, 2015 SCC Online Del. 8227; Cipla Ltd v. Novartis AG, 2017 SCC Online Del. 7393; Symed Labs v. Glenmark Pharma Ltd, 2015 SCC Online Del. 6745.
E.g., UPL Ltd v. Pradeep Sharma, 2018 SCC Online Del. 7315.
E.g., Koninklijke Philips NV v. Amazestore, 260 (2019) DLT 135; Telefonaktiebolaget LM Ericsson (Publ.) v. Intex Technologies (India) Ltd, 2015 SCC Online Del. 8229 (a final decree concerning SEPs); Koninklijke Philips NV v. Vivo Mobile Communications Co. Ltd, CS (COMM) 383 of 2020; Koninklijke Philips NV v. Xiaomi Inc., CS (COMM) 502 of 2020.
E.g., Shogun Organics Ltd v. Gaur Hari Guchhait, 263 (2019) DLT 516; Eisai Co. Ltd v. Satish Reddy, 2019 SCC Online Del. 8496.
E.g., F Hoffmann-La Roche Ltd v. Cipla Ltd, MIPR 2016 (1) 1; Koninklijke Philips, 260 DLT; Shogun Organics, 263 DLT (damages were awarded in the sum of about USD 25 million).
E.g., AstraZeneca AB v. Intas Pharmaceuticals Ltd, MANU/DE/1939/2020; B Braun Melsungen AG v. Rishi Baid, MANU/DE/0376/2009; Arif Abdul Kader Fazlani v. Hitesh Raojibhai Patel and Co., MANU/GJ/1304/2011; F Hoffmann-La Roche Ltd v. Cipla, 159 (2009) DLT 243 (DB).
AIR 1982 SC 1444, para. 17.
General Agreement on Tariffs and Trade, Oct. 30, 1947, 55 UNTS 194.
Panel Report, India – Patent Protection for Pharmaceutical and Agricultural Chemical Products, WTO Doc. WT/DS50/R (Sep. 5, 1997).
Appellate Body Report, India – Patent Protection for Pharmaceutical and Agricultural Chemical Products, WTO Doc. WT/DS50/AB/R (Dec. 19, 1997).
Panel Report, India – Patent Protection for Pharmaceutical and Agricultural Chemical Products, WTO Doc. WT/DS79/R.
WTO, Ministerial Declaration of 14 November 2001, WTO Doc. WT/MIN(01)/DEC/1, 41 ILM 746 (2002), para. 5 (“Accordingly, and in the light of paragraph 4 above, while maintaining our commitments in the TRIPS Agreement, we recognize that these flexibilities include: […] (b). Each Member has the right to grant compulsory licences and the freedom to determine the grounds upon which such licences are granted”).
WTO, Ministerial Declaration of 14 November 2001, WTO Doc. WT/MIN(01)/DEC/1, 41 ILM 746 (2002), para. 6.
Patents Act, 1970, §24B(1)(b).
Patents Act, 1970, §24B(1).
Patents Act, 1970, §24A(1).
Patents Act, 1970, §24A(2).
Patents Act, 1970, §24A(3).
Patents Act, 1970, §24D.
Patents Act, 1970, §60.
Patents Act, 1970, §2(1)(j).
Patents Act, 1970, §2(1)(ja).
Patents Act, 1970, §3.
Patents Act, 1970, §11-A.
Patents Act, 1970, §104-A.
Patents Act, 1970, ch. VI.
Patents Act, 1970, §86.
Patents Act, 1970, §107A(b).
Patents Act, 1970, ch. XIX.
The Tribunals Reforms (Rationalisation and Conditions of Service) Ordinance, 2021 (April 4, 2021).
Patents Act, 1970, §157-A.
The Tribunals Reforms (Rationalisation and Conditions of Service) Ordinance, 2021 (April 4, 2021).