
Pat-INFORMED: a new tool for drug procurement
By Mark F. Schultz, Professor, Southern Illinois University School of Law and Jaci Arthur, Director of Research, Institute for Intellectual Property Research, Illinois, USA
Developing a medicine is a highly complex process that involves numerous innovative steps, many of which can be protected with patents. While swallowing a small pill seems straightforward and far less complicated than, for example, using our smartphone, there is actually a great deal of specialist technology and expertise behind all the medicines we use. A huge amount of science and research goes into not only identifying an active ingredient to treat a condition, but also into calculating the right dosage and optimal release time – that is, how long the drug is active in our body.

It takes many years to develop a drug, and in the course of that development researchers may find that the molecules they are working with have new properties, attributes, and applications that also may be patentable. This means that many patents may be associated with any one drug or medicine. Differentiating between key patents that relate to a specific formulation of a drug and more peripheral patents covering niche applications can be difficult, particularly for a non-patent specialist, such as someone procuring drugs for an aid agency.
The challenge
The challenge of searching for drug patents is compounded by the fact that neither generic nor brand names for drugs are likely to appear in patents. Drug patents are most likely to be filed and granted early in the development of the product, long before the active ingredient of the formulation receives a generic name (i.e., an International Nonproprietary Name (INN)), or a registered trademark or brand name. The inevitable gap between terminology used to describe a drug in a patent and the name by which the product becomes generally known can create challenges for assessing the patent status of a medicine.

made difficult because of a gap in the terminology used to
describe a drug in a patent and the name by which the
product becomes generally known (photo: alvarez
/ E + / Getty Images).
A few countries have overcome this information gap by publishing databases, such as the United States’ “Orange Book,” which lists the patents that are relevant to each drug, according to the standard name for each medicine. The Orange Book also indicates when patents and other exclusivities expire. However, the Orange Book provides information only for the United States, and most countries do not provide such straightforward and simple references.
Determining the patent status of medicines made easy with Pat-INFORMED
The World Intellectual Property Organization (WIPO) and the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA), together with 20 participating global pharmaceutical companies, are teaming up to respond to this gap in information by effectively globalizing the concept of the Orange Book.
Under an initiative known as the Patent Information Initiative for Medicines (Pat-INFORMED), which was rolled out in September 2018, WIPO, IFPMA and its partners are providing information – specifically, patent numbers and application and grant dates – for approved medicines in jurisdictions around the world.
Pat-INFORMED links patent information worldwide with standard product names for important medicines. The database provides a clear, easy-to-use interface, where entering a product’s INN produces a concise list of patents, including the name of the company that owns the patents and the countries in which they have been granted.
Having this information available in one place is a big leap forward for all those interested in pharmaceutical patents. The primary goal of the project is to help procurement agencies obtain information on the patent status of a specific medicine in a particular jurisdiction. Pat-INFORMED will complement other patent database resources, such as those hosted by commercial entities, national patent offices, and WIPO’s own PATENTSCOPE, which currently holds around 71 million patent documents.
Why finding drug patents is a challenge
Most drugs are likely to have at least two names: a brand name chosen by the innovator and an INN (usually referred to as the generic name), which identifies pharmaceutical substances or active pharmaceutical ingredients. INNs are registered with the World Health Organization (WHO). For example, the INN of the well-known Hepatitis C drug that goes by the brand name Solvadi, is sofosbuvir. Similarly, the non-prescription pain reliever known widely by its INN, ibuprofen, also goes by brand names such as Advil, Motrin, Bufren, Nurofen, and many others.
INNs play an important role in public health by creating a shared nomenclature for drugs that cuts across national borders, brands, and health disciplines, simplifying communication and avoiding confusion. This shared language ensures that descriptions of drugs in prescriptions, medical records, purchase orders, inventories, and other communications are easily and consistently understood by all.
Doctors, health professionals, pharmacists, and specialists working in drug procurement use INNs as their common language (even if they also refer to brand names). INNs essentially comprise the word’s formulary of known medicines. There are more than 8,000 INNs registered with the WHO.
Unfortunately, the common language created by INNs does not extend to the language used in drug-related patents. Pharmaceutical patents frequently do not refer to INNs. They tend to describe a drug by its chemical composition or a name widely accepted by experts in the relevant field, but which differs from the INN.
The absence of INNs from patent documents is not a purposeful omission. It is a function of the point at which each arises within the development process. A patent application can easily be approved years before an INN application is submitted.
This gap in time between when patents and INNs are issued is a natural result of how the drug discovery process unfolds. Patents protect promising early breakthroughs in drug development. They precede, and may be a condition of, any further investment in testing and development. INNs are assigned once that promise is more developed.
By the time a pharmaceutical company files a request with the WHO for an INN, at least some relevant patents will have been filed, and likely granted, and the drug is likely to be about to start clinical trials. After the INN request has been filed, there is an average 15-month wait before the publication of a “Recommended INN,” followed by a four-month period during which objections to that name may be raised.
The inevitable absence of INNs from many relevant pharmaceutical patents means they do not share the language used by the rest of the healthcare community to identify drugs. It is therefore unlikely to be possible to identify relevant patent applications filed prior to the date on which the INN is issued through a search using the common, generic name. This can lead to confusion for those seeking to determine the patent status of a medicine. Inexperienced researchers may draw false confidence from incomplete results, while more seasoned ones face concerns that they may have missed something due to the lack of common nomenclature.
The challenge is particularly acute for non-patent experts working in the healthcare system who rely on INNs to identify patented medicines. Most procurement agencies employ healthcare specialists who are far more familiar with INNs than the chemical compositions, chemical names, or patent classifications used to search most patent databases. These difficulties are experienced across the healthcare system. Government ministries, researchers, aid groups, and others also often find it difficult to determine the patent status of drugs in different countries when searching by generic names.
Addressing a long-felt need
The difficulties associated with searching for patents using INNs have led many observers to consider alternative approaches. For example, the WHO has consistently noted the difficulties associated with this gap in language and has recommended various strategies to remedy the problem. WIPO’s Standing Committee on Patents has also assessed various options. India considered – but ultimately declined to impose – a requirement for disclosure of INNs in patent applications. Also, in 2016, a large ad-hoc coalition of civil society groups similarly called for more information linking drug patents to INNs.
Pat-INFORMED meets this long-felt need. Users can, for the first time, find information on all patents linked to an INN in just a few clicks.
Twenty leading pharmaceutical companies have committed to contributing to the database, which contains information on drugs on the WHO Essential Medicines List, as well as important oncologic, Hepatitis C, cardiovascular, HIV, diabetes, and respiratory therapy areas. The project aims to eventually cover all therapeutic areas and will explore the feasibility of including other treatments, complex therapies, and vaccines in the future.
Participation in Pat-INFORMED is voluntary. Its participants are committed to disclosing core patents in every country in which they are granted with details of all therapies covered. This information will be updated every six months for products on the Essential Medicines List and annually for all others.
This commitment, coupled with the comprehensive nature of the database, means that those researching the patent status of medicines now have access to a trusted resource that will make it easier to connect with patent owners. This is one of the initiative’s key objectives.
Drug procurement by ministries of health, manufacturers, donors, and aid agencies can be complex and costly. Poor drug procurement practices can even lead to shortages in high-quality, life-saving drugs. Therefore, new tools, like Pat-INFORMED, which facilitate and improve that process, are critical.
Companies participating in Pat-INFORMED
- AbbVie (USA)
- Astellas Pharma Inc. (Japan)
- Bristol-Myers Squibb (USA)
- Daiichi-Sankyo (Japan)
- Eisai (Japan)
- Gilead Sciences, Inc. (USA)
- GlaxoSmithKline (GSK) (United Kingdom)
- IPSEN (France)
- Johnson & Johnson (USA)
- Leo Pharma (Denmark)
- Lilly (USA)
- Merck KGaA (Germany)
- MSD (aka Merck & Co., Inc.) (USA)
- Novartis (Switzerland)
- Novo Nordisk (Denmark)
- Pfizer (USA)
- Roche (Switzerland)
- Shionogi Inc. (Japan)
- Takeda (Japan)
- UCB (Belgium)
With Pat-INFORMED, the language gap between INN and patent information will finally be closed. Importantly, the database is free, publicly accessible, and easy to use. Even a non-specialist can obtain better results, and more quickly, than previously possible.
A comprehensive, accurate, and user-friendly tool
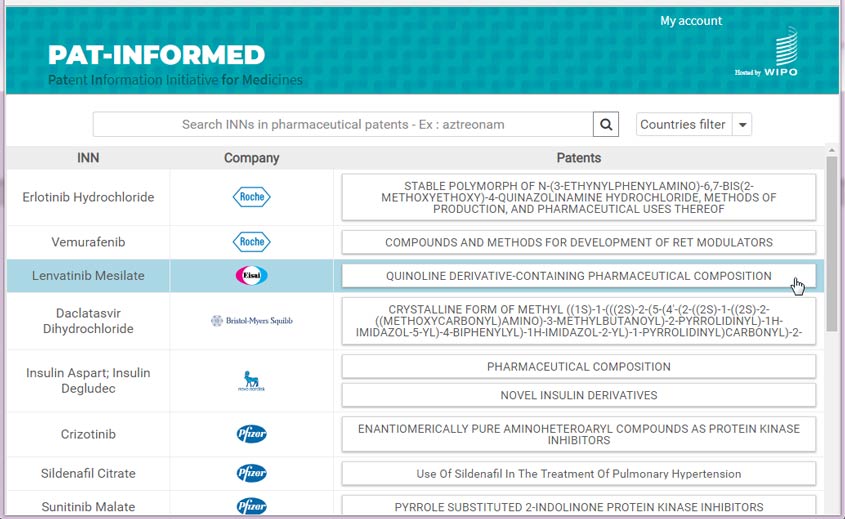
Pat-INFORMED breaks new ground in terms of its accuracy, its comprehensive coverage, and the way in which it consolidates and links to relevant information through a clear and intuitive interface. On accessing the database, users simply type in the INN or generic name of the drug about which they are seeking patent-related information. The results display all relevant INNs, with further groupings or families listed to the right of each one – Pat-INFORMED calls these groupings “cards.” By clicking on a “card,” the user can access key details of the relevant patent, a link to contact the company that holds that patent, and a separate box for each jurisdiction in which the patent is granted. Each of these boxes presents details of the patent, including its publication number, publication date, grant date, grant number, and, where available, a link to the text of the patent in WIPO’s PATENTSCOPE database (see Figure 1).
An indispensable tool for the global pharmaceutical market
Beyond hosting Pat-INFORMED, WIPO and IFPMA will act as intermediaries, ensuring that all bona fide requests received from procurement agencies are forwarded to the relevant pharmaceutical companies.
Crucially, all participating patent owners have committed to respond to such inquiries. Now, thanks to Pat-INFORMED, anyone involved in drug procurement seeking information on the patent status of drugs can find the right point of contact within a participating pharmaceutical company and is guaranteed a response. This makes Pat-INFORMED an indispensable tool for the global pharmaceutical marketplace.
Pat-INFORMED will help bridge the INN-patent information gap. While the database is not the sole source of information in drug procurement decision-making, it will facilitate the process of identifying the life-saving drugs that patients need, and will enable the drug procurement process to function more smoothly. The Pat-INFORMED database is already an important and critical advance for the patented medicines marketplace. It promises additional dividends in terms of supporting efficient drug procurement processes as other product areas are included and more companies join the initiative.
The WIPO Magazine is intended to help broaden public understanding of intellectual property and of WIPO’s work, and is not an official document of WIPO. The designations employed and the presentation of material throughout this publication do not imply the expression of any opinion whatsoever on the part of WIPO concerning the legal status of any country, territory or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. This publication is not intended to reflect the views of the Member States or the WIPO Secretariat. The mention of specific companies or products of manufacturers does not imply that they are endorsed or recommended by WIPO in preference to others of a similar nature that are not mentioned.